GLP-1 Agonists Market to Reach USD 170.75 Billion by 2033
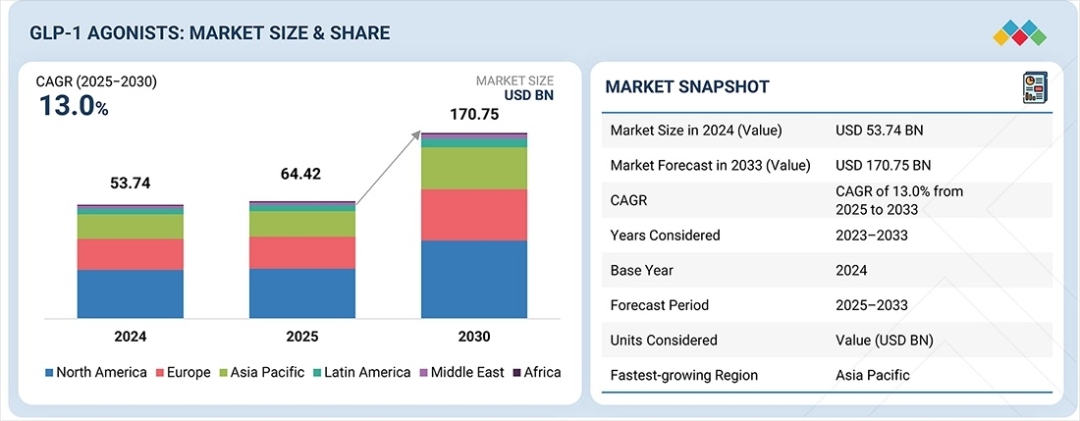
The global GLP-1 agonists market is on a trajectory of unprecedented growth, projected to soar from USD 64.42 billion in 2025 to a staggering USD 170.75 billion by 2033, representing a robust compound annual growth rate (CAGR) of 13.0%. This expansion is primarily fueled by the dual crises of global obesity and type 2 diabetes, which have reached epidemic proportions. As clinical evidence continues to mount regarding the cardiovascular and renal benefits of these therapies, GLP-1 analogues including blockbuster names like Ozempic, Wegovy, and Mounjaro are transitioning from niche diabetes treatments to foundational pillars of metabolic health. The market is further energized by a shift toward more convenient administration formats, such as tablets and long-acting injectables, which are broadening patient access and improving long-term adherence.
Download PDF Brochure:https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=218746186
Strategic Expansion of Manufacturing Capacities and Indication Diversification
To meet the skyrocketing global demand that has frequently outpaced supply in recent years, major pharmaceutical players are aggressively expanding their manufacturing infrastructure. This includes significant investments in dedicated facilities for active pharmaceutical ingredients (APIs) and finished dosage forms. Beyond simple capacity, the market is being redefined by "indication expansion." Regulatory bodies are increasingly approving GLP-1 agonists for conditions beyond glycemic control, including cardiovascular risk reduction and noncirrhotic metabolic dysfunction-associated steatohepatitis (MASH). For instance, recent FDA approvals for Wegovy to treat liver fibrosis and reduce major adverse cardiovascular events (MACE) have opened massive new patient populations. This diversification ensures that GLP-1 therapies are no longer confined to the diabetes specialty but are integrated into comprehensive chronic disease management protocols.
Technological Breakthroughs in Oral Formulations and Multi-Agonist Molecules
The next frontier of the GLP-1 market lies in the move from painful weekly injections to user-friendly oral delivery systems. While Rybelsus currently stands as the primary oral option, the industry is closely watching the pipeline for next-generation candidates like Orforglipron, expected to hit the market by 2027-2028. These oral advancements eliminate "needlephobia," a major barrier to treatment initiation, and simplify logistics by removing the need for cold-chain storage. Simultaneously, the emergence of multi-agonists is disrupting the status quo. Drugs like Mounjaro and Zepbound (tirzepatide) utilize a dual-mechanism approach, activating both GIP and GLP-1 receptors to achieve superior weight loss and glucose monitoring compared to single-hormone therapies. Even more potent triple-agonists, such as Retatrutide, are currently in Phase 3 trials, promising even more dramatic clinical outcomes that could eventually rival bariatric surgery.
Regional Dominance of North America and Rapid Growth in Asia Pacific
From a geographic perspective, North America continues to hold the largest market share, bolstered by a highly structured healthcare ecosystem, high per-capita healthcare spending, and early adoption of innovative formulations. The region serves as the primary hub for clinical trials and rapid product launches, supported by favorable reimbursement scenarios for diabetes. However, the Asia Pacific region is projected to register the highest CAGR through 2033. This surge is driven by a massive undiagnosed patient pool in countries like China and India, coupled with rapidly improving healthcare infrastructure and increasing government focus on metabolic disease management. As global pharma companies like Novo Nordisk and Eli Lilly localize their strategies and as domestic biosimilar manufacturers enter the fray, the accessibility of GLP-1 therapies is expected to expand across emerging economies.
Request Sample Pages-https://www.marketsandmarkets.com/requestsampleNew.asp?id=218746186
Navigating Market Challenges: Payer Controls and the Rise of Biosimilars
Despite the optimistic forecast, the market faces significant hurdles, most notably stringent cost and payer controls. High price points have led insurers to implement restrictive eligibility criteria and favor single-drug formularies. This financial strain is creating a unique market opening for biosimilars. With patent expiries for major molecules like semaglutide looming as early as 2026, the industry is preparing for a wave of affordable alternatives. Companies such as Teva Pharmaceuticals and Biocon are positioned to disrupt the market by offering cost-effective versions of these life-changing drugs. Furthermore, the industry must contend with the "gray market" of compounded and counterfeit products, which pose serious safety risks. Maintaining market integrity through tighter regulatory oversight and patient education will be crucial as the GLP-1 agonists market evolves into a nearly USD 171 billion global powerhouse by the end of the decade.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: GLP-1 Agonists Market to Reach USD 170.75 Billion by 2033
Information contained on this page is provided by an independent third-party content provider. XPRMedia and this Site make no warranties or representations in connection therewith. If you are affiliated with this page and would like it removed please contact [email protected]